Lung cancer: Everything you need to know
What is lung cancer?
Definition
Lung cancer is the leading cause of cancer deaths. It mainly affects men but is increasing significantly among women. The main risk factor identified is active or passive smoking (also called second-hand smoke).
Also known as bronchopulmonary cancer or bronchial cancer, lung cancer develops mainly from cells in the lining of the bronchi and bronchioles (ducts that carry outside air to the lungs). More rarely, it is possible that the cancer develops in the alveoli (small sacs where gas exchanges with the blood take place) or in the pleura (membrane surrounding the lung).
There are different types of lung cancer:
- Non-small cell lung cancer (NSCLC) which accounts for 85% of lung cancers. These include:
- Adenocarcinoma (about 45% of cases)
- Squamous cell carcinoma (around 25% of cases)
- Large cell (undifferentiated) carcinoma (about 10% of cases)
- And other, much rarer forms (carcinoid tumours, etc.)
- Small cell lung cancer (SCLC) which accounts for 15% of lung cancer.
How common is lung cancer?
Lung cancer is the leading cause of cancer deaths in the UK and worldwide.
In the UK, it is the 3rd most common cancer, after breast cancer and prostate cancer. It is the 2nd most common cancer for both men and women.
It is estimated that 47,000 new cases of lung cancer in the UK are diagnosed every year and 25,100 deaths occur.
75% of lung cancer cases are diagnosed at an advanced stage. The average age at diagnosis is 67 years old for men and 65 years old for women.
1 in 13 men and 1 in 15 women will be diagnosed with lung cancer in their lifetime but this gap tends to decrease due to the increase of smoking in women.
Life expectancy at 5 years is estimated at 21% and at 10 years after diagnosis, it is estimated at 10%.
Symptoms and complications of lung cancer
In the early stages of the disease, there may be no symptoms or symptoms that seem ordinary in a smoker or ex-smoker.
However, at an advanced stage, various respiratory symptoms may be observed and require consultation with a GP:
- Cough that persists and tends to get worse, with no apparent cause
- Recent onset of shortness of breath and difficulty breathing
- Chest pain that worsens with the cough
- Sputum (phlegm) containing blood (haemoptysis)
- Hoarseness of the voice that does not pass
- Wheezing while breathing
- Repeated lung infections (bronchitis, pneumonia, etc.).
Other, more general, symptoms may appear depending on the extent of the tumour (presence of metastasis or not):
- Abnormal fatigue (asthenia)
- Difficulty swallowing (dysphagia)
- Rapid loss of appetite and weight
- Oedema (swelling) of the face or neck
- Prolonged fever and headache
- Bone pain...
Causes and risk factors of lung cancer
There are 2 main risk factors for the development of lung cancer:
- Smoking (active or second-hand): responsible for 85 to 90% of lung cancers.
Indeed, cigarette smoke contains more than 7000 chemical components, 60 of which are recognised as carcinogenic (polycyclic aromatic hydrocarbons, nitrosamines, benzene, formaldehyde, radioactive compounds such as polonium, etc.). These components are carcinogenic by numerous mechanisms such as binding to DNA inducing mutations, oxidative stress, inflammation or epigenetic modifications (modifications of gene expression without altering the DNA).
The risk of lung cancer is considered to increase both with the amount of tobacco smoked and the length of time a person has smoked.
- Occupational and environmental exposure to toxic components
Asbestos, in particular, increases the risk of lung cancer fivefold in a non-smoking patient and fiftyfold in a smoking patient.
Numerous chemical elements (cobalt, chromium, cadmium, arsenic, nickel, silica, radon, etc.), exposure to ionising radiation, polycyclic aromatic hydrocarbons (PAHs) and diesel exhaust are also involved in the development of lung cancer.
Finally, chronic respiratory diseases (COPD, emphysema, tuberculosis, etc.), inhaled cannabis and the consumption of beta carotene, in the long term and in high doses (as shown in the study conducted by Françoise Clavel-Chapelon of the INSERM-Institut Gustave Roussy), by smokers or people exposed to asbestos, significantly increase the risk of developing lung cancer.
Diagnosing lung cancer
Systematic diagnostic examinations
Clinical examination: The patient is interviewed by the GP in order to look for risk factors (smoking, exposure to toxins, etc.) and any changes in general health.
The doctor will examine the patient, tap on his or her chest (percussion) and listen to the sounds of the lungs and heart with a stethoscope, palpate the neck and the area above the clavicles to look for swollen lymph nodes, palpate the abdomen to assess the size of the liver, measure the blood pressure and pulse, and calculate the BMI (Body Mass Index).
Biological exams: With the help of a blood test, an estimation of kidney function is carried out before the injected CT. A haemostasis assessment, with the measurement of the prothrombin rate (PT), the activated partial thromboplastin time (aPTT) and the measurement of platelets, may be prescribed to facilitate subsequent care (with a view to taking samples for histological purposes).
Chest X-ray: Performed from the front and profile, the aim of this exam is to reveal the presence of abnormalities within the lungs, using an X-ray machine. However, the X-ray cannot distinguish whether the abnormality observed is benign or malignant. It may also be normal even when lung cancer is present (particularly for small tumours).
Chest CT or computerised tomography (CT): This requires the injection of a contrast agent (often iodine) in the absence of contraindications (kidney failure). This exam allows you to perform a series of images of the lung in horizontal slices. It makes it possible to locate or not the presence of an abnormality, as well as its size and location. However, the scanner gives no information on the type of cancer cells.
Bronchoscopy with biopsy: The biopsy is often performed during a bronchial fibroscopy. This involves inserting a thin, flexible tube, connected to a light and a micro-camera, which, when inserted through a nostril, allows the inside of the trachea and bronchi to be observed.
A pathological and/or cytological examination is carried out on tumour fragments taken by biopsy. This examination, carried out with the naked eye and then under a microscope by a specialist, confirms the diagnosis of lung cancer. It provides precise information on the type of cancer (small cell or non-small cell), looks for molecular changes (EGFR gene mutation or ALK or ROS translocation, etc.) and, if performed after surgery, estimates the stage of the cancer.
When a fibroscopic biopsy is not feasible, the medical team may take a tumour or pleural fluid sample through the chest wall, or a lymph node sample, under local anaesthetic. Tissue sampling can also be performed during a surgical exploration, requiring a short hospital stay.
Cancer staging examinations
MRI with contrast: Systematic in the case of non-small cell lung cancer (NSCLC), it allows precise images of the brain to be created and thus to search for the presence of lesions or brain metastasis.
Positron emission tomography (PET, PET-CT, PET Scan, or Petscan): A painless examination that produces cross-sectional images of the whole body after a tracer (a weakly radioactive product) has been injected into the blood. This tracer has the particularity of binding to cancer cells and thus allows the search for extra-cerebral lesions or metastasis.
Abdominal CT scan: Performed at the same time as the thoracic CT scan, it allows the exploration of the liver and adrenal glands.
TNM Classification
This classification is necessary in order to choose the treatment strategy.
- T = Size or direct extent of the tumour:
- TX: Tumour of unknown primary or tumour proven by the presence of malignant cells in bronchopulmonary secretions but not visible on radiological and endoscopic exams.
- T0: No evidence of tumour.
- Tis: Carcinoma in situ (highly localised cancer).
- T1: Tumour of 3 cm or less in its largest dimension, surrounded by lung or visceral pleura (membrane surrounding the lung), with no evidence of invasion further upstream of the lobar bronchi on bronchoscopy (i.e. not in the stem bronchi):
- T1a(mi): Minimally invasive adenocarcinoma
- T1a: ≤ 1 cm
- T1b: > 1 cm and ≤ 2 cm
- T1c: > 2 cm and ≤ 3 cm.
- T2: Tumour larger than 3 cm, but 5cm or less, or with one of the following:
- invasion of a primary bronchus regardless of its distance from the carina (division of the trachea into left and right primary bronchi), but
- without invasion of the carina
- invasion of the visceral pleura
- atelectasis (collapse or retraction of the pulmonary alveoli) or obstructive pneumonia
- T2a: > 3 cm but ≤ 4 cm
- T2b: > 4 cm but ≤ 5 cm.
- T3: Tumour greater than 5 cm and 7 cm or less or associated with separate tumour nodule(s) and in the same lobe, or directly invading:
- the chest wall (including vertex tumours)
- the phrenic nerve
- the parietal pleura or parietal pericardium.
- T4: Tumour larger than 7 cm or associated with separate lung nodule(s) or directly invading: the mediastinum, heart or great vessels, trachea or carina, diaphragm, recurrent nerve, oesophagus or vertebral bodies.
- N = Degree of spread to regional lymph nodes:
- Nx: Lymph nodes cannot be assessed.
- N0: No regional lymph nodes metastasis.
- N1: Regional lymph node metastasis present, at some sites tumour spread to closest or small number of regional lymph nodes.
- N2: Tumour spread to an extent between N1 and N3.
- N3: Tumour spread to more distant or numerous regional lymph nodes.
- M = Presence of distant metastasis:
- M0: No distant metastasis.
- M1: Metastasis to distant organs (beyond regional lymph nodes):
- M1a: Separate tumour nodule(s) in a contralateral lobe, or pleural nodules or malignant pleurisy or malignant pericarditis
- M1b: Single extra-thoracic metastasis in a single organ
- M1c: Multiple extrathoracic metastases in a single or multiple organs.
Stages of lung cancer
The TNM classification thus makes it possible to differentiate several stages of lung cancer:
- Localised cancer (stages I and II):
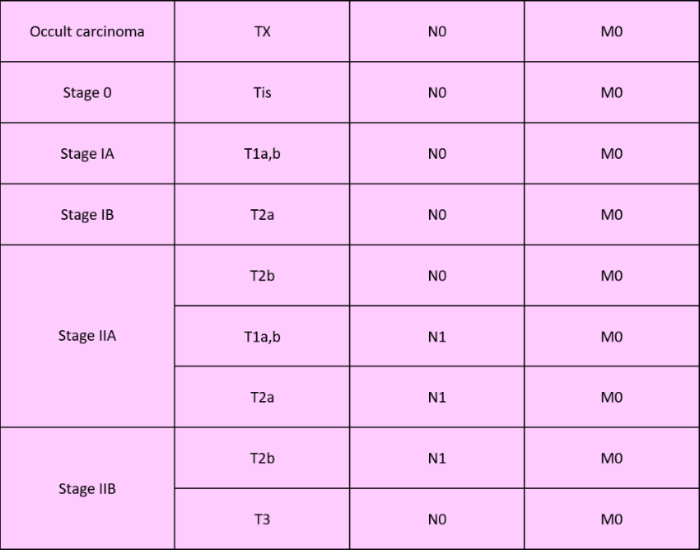
- Localised cancer (stage III):
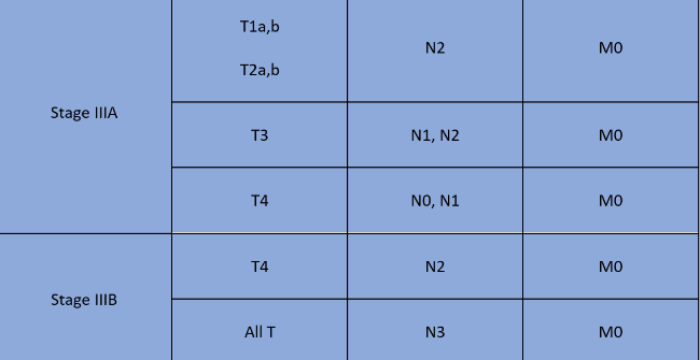
- Metastatic cancer (stage IV):

Pre-therapeutic assessment
During a multidisciplinary consult, a pre-therapeutic assessment is conducted. The assessment identifies comorbidities and evaluates the feasibility of the different treatment options. It includes an evaluation of:
- Patient performance status (PS):
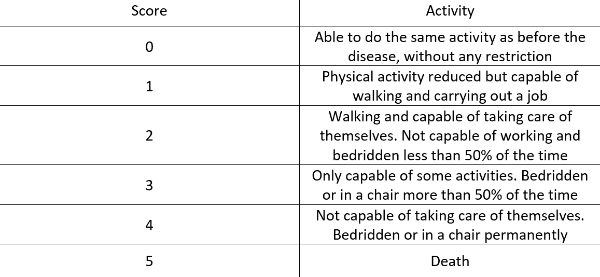
Source: Guide du parcours de soin : Cancers broncho-pulmonaires, HAS; Institut national du cancer
- BMI, equal to weight (kg) divided by height squared (m2), and the detection of weight loss in the previous 3 months
- Smoking, as well as encouragement and support for smoking cessation.
Lung cancer treatments
The treatment of lung cancer differs according to the type (NSCLC or SCLC) and stage (stage I, II, III or IV) of the cancer.
The different treatment options are:
•Surgery, if possible
•Chemotherapy
•Radiotherapy
•Targeted therapies
•Immunotherapy
•Symptomatic treatments (which is called "best supportive care").
Non-small cell lung cancer (NSCLC)
Operable cancer (stages I and II): Surgery is the standard treatment. It consists of removing part or all (rarely) of the lung where the tumour is located, as well as the surrounding lymph nodes (hilar and mediastinal lymph node dissection). In some cases, conventional chemotherapy is performed before (known as neoadjuvant chemotherapy) and/or after surgery (if the tumour is ≥ 4 cm or if there is lymph node involvement).
Cancer localised in the chest but inoperable (stages I and II): Stereotactic radiotherapy, which allows high-dose irradiation of very small volumes, is the preferred technique. In some cases, conformal radiotherapy, which destroys cancer cells with radiation while preserving as much healthy tissue as possible, may be offered, either alone or in combination with chemotherapy. Chemotherapy alone may also be considered. Finally, in certain situations, thermo-ablation (destruction of the tumour by cold or heat) is performed.
Locally advanced unresectable cancer (stage III): Treatment consists of conventional chemotherapy combined with radiotherapy. In case of contraindication to the combination of the two, each of these techniques, alone, can be considered. In addition, when immunohistochemistry (IHC) detects >1% of programmed cell death ligand-1 (PD-L1) protein, which allows tumours to escape the immune system, durvalumab (Imfinzi®) immunotherapy is used as consolidation treatment for 1 year.
Metastatic cancer (stage IV): Surgery is not offered as first-line treatment for metastatic lung cancer. Its management is based solely on drug approaches or radiotherapy.
The type of treatment will depend on whether the NSCLC is non-squamous (mostly adenocarcinoma) or squamous.
In non-squamous histology, the search for oncogenic changes is the first step, with at least the need for EGFR genotyping, and the search for ALK, ROS1 and BRAF rearrangements.
In case of oncogenic changes, different targeted therapies can be used in the first line:
- osimertinib, afatinib, gefitinib, erlotinib if there is EGFR mutation,
- alectinib, brigatinib, ceritinibif there is ALK translocation,
- crizotinib if there is ROS 1 translocation,
- dabrafenib + trametinib (as second line treatment) if there is a BRAF V600 mutation.
In the absence of oncogenic alterations, the key biomarker is PD-L1, expressed by tumour cells. The choice of treatment will then depend on:
- he level of PD-L1 expression (≥ 50%)
- the patient's condition (PS 0-1, 0-2, or 3-4);
- the patient's age ≥ or < 70 years.
Thus, if PD-L1 ≥ 50% and the performance score (PS) is 0-1, immunotherapy with pembrolizumab (Keytruda®) alone is used as first-line therapy. If tumour progression is observed, platinum salt chemotherapy is performed.
Regardless of PD-L1 expression level and if PS is 0-2, the combination of pembrolizumab immunotherapy with pemetrexed and platinum salt chemotherapy is typically used.
In patients with impaired general condition, with a PS ≥ 2, carboplatin-based chemotherapy alone, often combined with pemetrexed, is the standard treatment. Finally, in the absence of immunotherapy and contraindication, targeted therapy with bevacizumab can be added to chemotherapy, such as carboplatin + paclitaxel or cisplatin + gemcitabine.
If there is tumour progression with a PS of 3 to 4, doctors resort to palliative care.
In the situation of squamous cell histology, the search for oncogenetic changes is not relevant, except for non-smokers or light smokers (< 15 pack-years). PD-L1 remains a key biomarker to guide treatment:
- If PD-L1 is ≥ 50%: immunotherapy with pembrolizumab monotherapy is used;
- Otherwise, chemotherapy remains a standard treatment.
Note that permetrexed and bevacizumab are not indicated for non-squamous NSCLC.
Small cell lung cancers (SCLC)
In the early stages, radiotherapy and concurrent chemotherapy are used:
At an early stage, radiotherapy and chemotherapy are used in combination:
- Chemotherapy is most common: platinum salt-based (cisplatin or carboplatin) combined with etoposide.
- A complete response will be followed by prophylactic cranial radiotherapy.
However, the majority of patients are diagnosed at metastatic stage (stage IV). In this situation, immunotherapy with atezolizumab or durvalumab in combination with platinum-based chemotherapy and etoposide are used.
Living with lung cancer
Medical follow-up
Close medical follow-up is necessary for several years in patients who have received treatment for lung cancer, in order to monitor possible early complications (such as febrile aplasia during chemotherapy) or late complications (such as cisplatin-related peripheral neuropathy) and to prevent the occurrence of cancer relapse.
This follow-up is carried out by a multidisciplinary team consisting of, among others, a general practitioner, a pulmonologist, an oncologist, a radiologist, a surgeon, a nutritionist or a psychologist.
It is usually based on consultations and chest X-rays every 3 months for 2 years, then every 6 months for 3 years. CT scans and other complementary examinations are also carried out every 6 months for 2 years, then every year for 3 years.
A healthy lifestyle
First of all, it is preferable to stop smoking for good, because continuing to smoke after treatment increases the risk of complications from the treatments and increases the risk of a secondary cancer.
In order to combat the progression of the cancer and the undesirable effects of the treatments, it is also important to maintain a varied and well-portioned diet. In particular, it is important to opt for foods with anti-inflammatory properties and rich in antioxidants: fruit and vegetables, olive oil, oily fish, nuts, etc.
Finally, staying active and engaging in regular physical activity is important for the body and the morale. Walking, gardening or cycling can help combat fatigue after treatment.
Psychological support
It is important to communicate, to share doubts and fears about the disease (appearance of new side effects, questions about a new treatment) with the doctor treating you, but also with your loved ones. It is also beneficial to turn to discussion groups and organisations for patients living with the same disease. You can also join online patient communities such as Carenity's Lung Cancer Forum. If necessary, psychological support (psychologist, psychiatrist, etc.) can be considered.
In case of difficulties in daily life, it is possible to ask for home help to carry out daily activities (getting up, washing, eating, domestic activities, administrative procedures, organisation of family life, etc.).
If difficulties occur in daily life, it is possible to ask for help at home to carry out the daily tasks (getting up, washing, food, domestic activities, administrative tasks, organising family life, etc.).
Returning to work
A leave of absence from work, the duration of which depends on the type of treatment and possible post-operative complications, may be prescribed by the patient's doctor. In addition, appropriate return-to-work adjustments can be planned, with the implementation of a part-time regimen over a short period or an adaptation of the workstation or office space, for example. Finally, after a period of more than 30 days off work, a pre-work visit to the occupational physician is recommended.
In conclusion, lung cancer is the leading cause of cancer death and the second most common cancer, all sexes combined. It is usually diagnosed late, in advanced stages, and the main risk factor is smoking. There are different therapeutic approaches (chemotherapy, radiotherapy, targeted therapies, etc.) and the choice of treatment depends mainly on the type of cancer (non-small cell lung cancer NSCLC in 85% of cases and small cell lung cancer SCLC in 15% of cases).
Lung cancer community
Breakdown of 784 members on Carenity
Average age of our patients
Fact sheets
See more